FDA’s Moderna decision demonstrates how science and government are incompatible

Photo Credit: Getty
Last week, the FDA announced that it would not be considering Moderna’s application for a new flu vaccine because the control group Moderna used was inadequate. This announcement has sent shockwaves through the drug development and scientific communities as it is an escalation of the Trump administration’s skepticism toward a new approach to vaccine development. The decision highlights several problematic aspects of the FDA approval process and regulation more broadly, and it may stymie promising potential future medicines.
Background
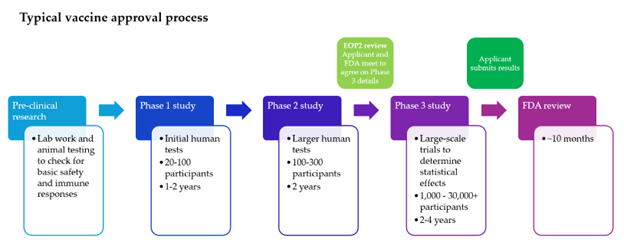
In the case of the Moderna vaccine, the process kicked off in 2021. The End-of-Phase 2 meeting, held prior to the Phase 3 study where the FDA and applicant come to an agreement on the structure of the trial, took place in April 2024. At that time, the FDA approved Moderna’s trial and endpoint. However, upon preliminarily reviewing the results of the Phase 3 trial last week, the FDA now concluded that the control group was inadequate to demonstrate efficacy of the drug, contrary to the 2024 agreement.
To be more precise, the original agreement was that Moderna would compare its vaccine to a “standard dose” flu vaccine, to show that their new version would beat the standard. The FDA argues that Moderna should have compared it to a high dose flu vaccine.
In one sense, the FDA is correct. To properly test the efficacy of a new treatment, it should be compared to the current standard treatment. With flu vaccines, the current standard treatment is a standard dose (15 micrograms of the antigen) for those under 65, and a high dose (60 micrograms) for those over 65.
The FDA agreed, however, that the standard dose would be acceptable for all ages for this trial. In its Refusal-to-File letter, the FDA has reconsidered and now believes the standard dose control is insufficient. Consequently, the FDA will not review the results any further.
As with many political flips, one could make the case that it was the 2024 FDA that put politics ahead of science by relaxing the standard for comparison to favor mRNA technology while today’s FDA is merely putting science ahead of politics, as it should. The reverse can also be argued as the Trump administration has been highly skeptical of mRNA-based science and vaccines and may have found an excuse to slow it down.
There’s truth to both of these arguments, and this is an important reason why government regulations should be as minimal as possible. Too often with regulations, the decisions become arbitrary and contingent on the politics or personal opinions of those in charge. Pure technocracy and science-based governance are unattainable, and the only solution is to reduce government oversight of decisions.
Combining a highly polarized country, a highly regulated economy, and a bureaucracy that has little accountability creates a situation where rules and regulations are erratic and unpredictable, which is destructive for economic growth and progress. The FDA’s decision is only the latest instance of that. Tariffs were the story of 2025, and the FTC underwent a similar flip during the Biden administration.
What this decision should demonstrate is that a regulatory state ruled by the discretion of political appointees is fundamentally fragile and harmful to an economy.
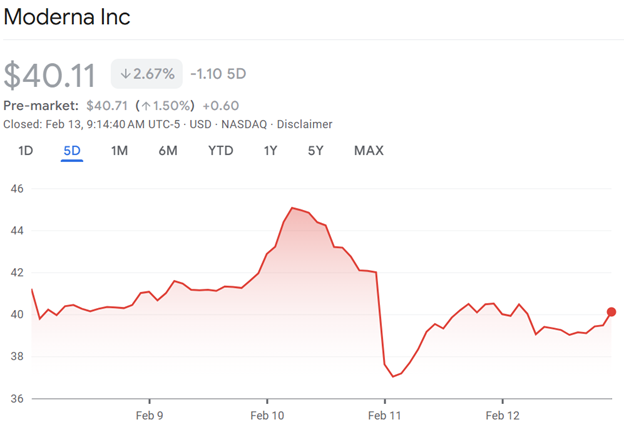
In the aftermath of the FDA’s surprise announcement, Moderna stock dropped 12%, erasing nearly $2 billion from its market value.
Efficacy standard
The decision also underscores questions about the efficacy standard itself. The FDA evaluates two aspects of new drugs: safety and efficacy. The FDA requires developers to prove that the drug is both safe for use and accomplishes its goal before approval.
The efficacy requirement adds complexity and costs to the drug approval process. The Moderna case highlights one such complication: determining the correct control group. As it currently stands, the FDA requires a new drug to be more effective than the current best treatment, but this will tend to limit the number of treatments available. Alternative approaches would be to allow treatments that perform worse than the current best treatment but better than a standard treatment, or better than a placebo. Or perhaps some treatments work for some people better than others, which would require more approvals, assuming there was a large enough population to meet the current FDA thresholds. Although the FDA accounts for some of this nuance, doing so increases costs on both sides of the process, reduces the availability of medicines, and limits competition among different treatment options in favor of an increasingly inappropriate and outdated one-size-fits-all standard.
The upshot is that the FDA’s efficacy standard should be reconsidered, and potentially removed altogether as a requirement for approval.
mRNA vaccines hold significant promise
While mRNA vaccines are highly politicized, they hold a lot of potential both for the flu and other viruses. For the flu, the current prevailing technology of developing flu vaccines takes approximately six months. Because the flu virus is always mutating and changing, by the time the vaccine is ready for deployment, the virus has moved on, and the vaccine loses some of its efficacy.
Source: CDC
By contrast, an mRNA vaccine can be developed in one-third of the time. Faster development would mean less time for the virus to change, higher efficacies, and fewer flu deaths.
Not to mention other viruses that mRNA may be able to vaccinate against. Late last month, Moderna announced that they would stop development in the US of other vaccines in light of the new skepticism of vaccines.
Politicized science
The real losers from the FDA’s decision are not Moderna, the drug industry, or mRNA advocates, but Americans – and likely everyone else given the importance of the US market. The new technology for vaccines has a significant potential to revolutionize the entire vaccination process, creating vaccines that prevent more diseases and improve effectiveness against the ones already targeted. Millions of Americans want access to this new technology. Because of politics, the FDA is holding up progress and withholding these developments from people who want them.