Last Chance for the 115th: Protect Lifesaving Vaping Products

This June here at OpenMarket we’ll be looking at what the 115th Congress, which began January 3, 2017 and runs through January 3, 2019, has accomplished so far and what might still be achieved for limited government and free markets before it’s over. Read more about the Competitive Enterprise Institute’s recommendations for legislative reform here.
 In CEI’s Free to Prosper: A Pro-Growth Agenda for the 115th Congress, we made the case that Congress must act before anti-tobacco zealots in and outside of government eliminate life-saving vaping products. To date, Congress has failed to take action. The nearing end of the 115th Congress may very well prove to be the last chance for those members who truly care about public health to correct this grave error before it is too late.
In CEI’s Free to Prosper: A Pro-Growth Agenda for the 115th Congress, we made the case that Congress must act before anti-tobacco zealots in and outside of government eliminate life-saving vaping products. To date, Congress has failed to take action. The nearing end of the 115th Congress may very well prove to be the last chance for those members who truly care about public health to correct this grave error before it is too late.
In 2009, Congress enacted the Family Smoking Prevention and Tobacco Control Act (TCA), which vested the U.S. Food and Drug Administration (FDA) with the authority to regulate tobacco products. In 2014, the FDA announced—without any direction from Congress—that it would begin regulating all packaged nicotine products as tobacco under the TCA. By lumping safer nicotine products, like e-cigarettes, in with traditional cigarettes, which kill upwards of half their users, this so-called “deeming rule” threatens to kill the most promising development in the realm of tobacco harm reduction we have ever seen.
Last summer, FDA Commissioner Scott Gottlieb delayed the deeming rule, but that only gives vapor products a reprieve from extinction until 2022. After that, all e-cigarettes will have to exit the market until they can get pre-market approval from the FDA—a process estimated to cost $1 million for each product with no guarantee the agency will approve any applications. In fact, to date the FDA has only approved a handful of products—all made by the Swedish snus company Swedish Match (snus is a moist tobacco chew product).
Traditional cigarettes do not need to acquire this pre-market approval. A provision in the law allows products that were on the market, or are substantially similar to products on the market, prior to 2007 to skip this onerous process. E-cigarettes, on which thousands of ex-smokers rely, do not qualify for this exemption since no e-cigarettes were available on the U.S. market before 2007.
The vast majority of e-cigarette and e-liquid manufacturers are small, independent companies, so it is likely that most will not even attempt to acquire FDA approval. By the agency’s own estimate, this will eliminate 99 percent of the currently available e-cigarette products from the market. Yet, it leaves traditional cigarettes relatively unscathed, a situation that should worry anyone who truly cares about public health.
E-cigarettes are a net benefit to public health. Despite the concerns aired by anti-smoking advocates, e-cigarettes have not led to a rise in adolescent smoking or adult smoking. In fact the rise of e-cigarettes’ popularity corresponded to a significant acceleration in the decreasing smoking rate.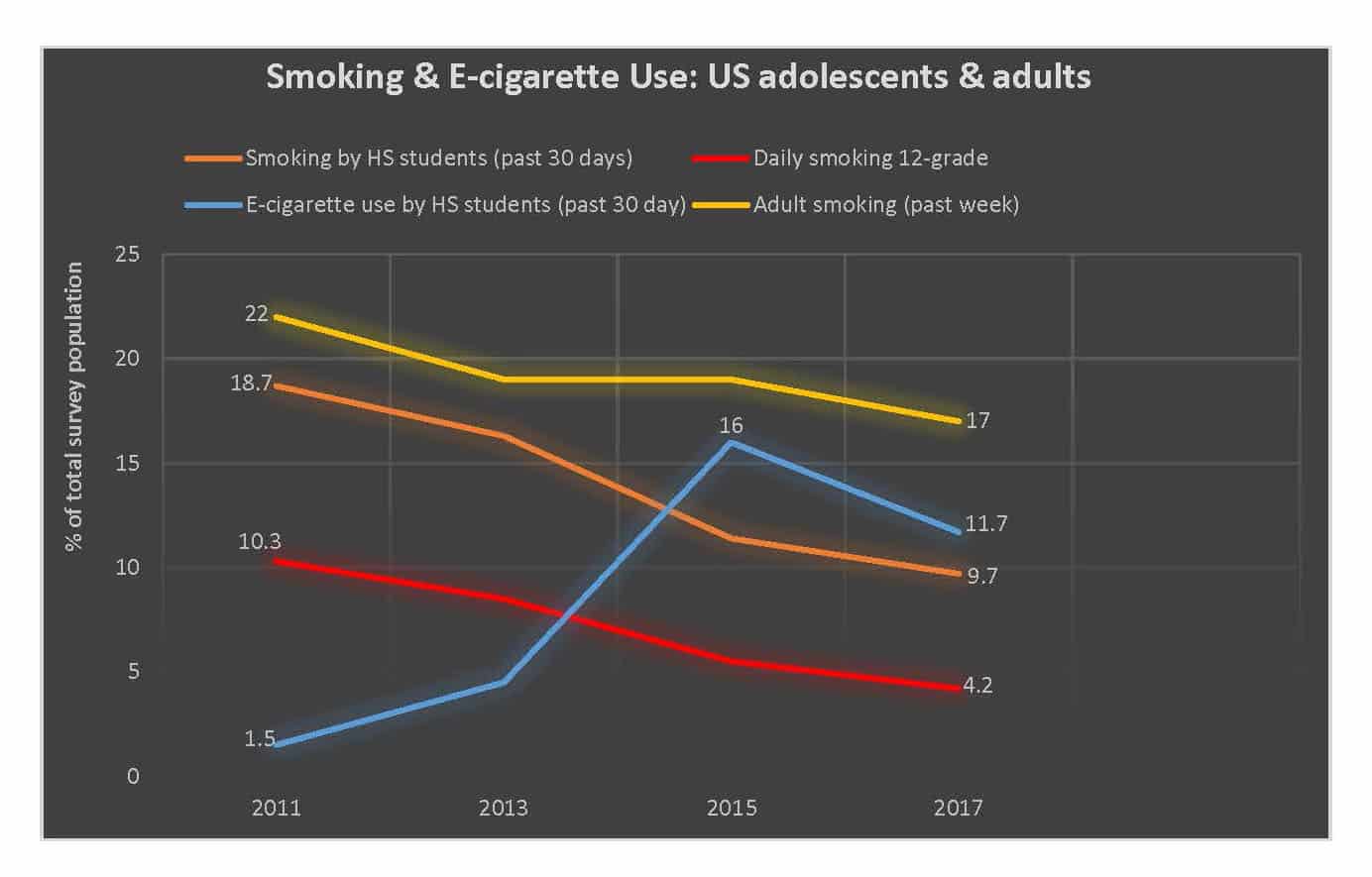
This rise apparently peaked in 2015. Since then, the U.S. Centers for Disease Control and Prevention (CDC) has documented an astounding 30 percent decline in self-reported use of e-cigarettes by high schoolers and a nearly 40 percent drop among middle schoolers. Even the 11.7 percent of high schoolers who reported vaping at least once in the last month is an exaggeration. At the height of its popularity (2014-2015), studies found that only 2 percent of high schoolers reported vaping on a daily basis, with just 8 percent vaping three or more times a month. Furthermore, surveys examining which teens vape found that just 22 percent of 12th graders and 13 percent of middle schoolers said they used e-cigarettes with nicotine.
Based on CDC’s latest population studies, this means that, at worst, less than .0015 percent of middle school and high school students are regularly vaping nicotine. This is certainly not an “epidemic” worthy of legislation to make e-cigarettes less attractive. Certainly, it does not warrant legislating e-cigarettes, which help adult smokers quit smoking, out of existence.
In almost every other realm of public health, experts recognize the folly of an abstinence-only approach. But, while we know that restrictions on vaping lead to increasing youth smoking and smoking among pregnant women, anti-vaping advocates do not seem to care about these realities. In other words, they are more comfortable with the idea of trapping smokers in their deadly addiction than allowing them the freedom to assess the risks and benefits of vaping and make their own choices about their health.
This is why Congress must step in and protect these harm-reducing tobacco alternatives from anti-vaping zealots.
When Congress enacted the TCA in 2009, it not only gave the FDA oversight over tobacco products, but also instructed the agency to promulgate rules that “promote and encourage the development of innovative products and treatments (including nicotine-based and non-nicotine-based products and treatments)” in order to achieve:
- Total abstinence from tobacco use;
- Reductions in consumption of tobacco; and
- Reductions in the harm associated with continued tobacco use.
Instead, the agency promulgated rules that would do the exact opposite: eliminating innovative products on which smokers already use to reduce or end their smoking habit. Clearly, this was not what Congress intended when it passed the TCA and runs counter to the FDA’s purported goal of protecting public health.
What Congress can do:
- Amend the Tobacco Control Act to direct the FDA to create a “file and use” approval process for tobacco products that are demonstrably less harmful or can be reasonably assumed to have a net positive effect on public health. This would require makers of vaping products to submit ingredient and safety disclosures to the agency and reserve the agency’s ability to regulate such products, but not force them to wait for prior approval before bringing products to market;
- Amend the TCA to allow less harmful nicotine products to be advertised as such;
- Modify the TCA’s “predicate” date (the grandfather date) to 2016 so that, at least, the products currently available to consumers can remain on the market; and
- Reject proposals that eliminate e-cigarette flavors or restrict sales to adults over the age of 21.
All reasonable experts now agree that e-cigarettes, if not risk-free, are significantly less harmful than cigarettes, exposing users to 95-99 percent fewer harmful and potentially harmful chemicals. Some studies have found that smokers who attempt to quit using e-cigarettes are 60 percent more likely to succeed than those using over-the-counter nicotine replacement therapies or willpower alone. And researchers recently estimated that getting smokers to switch to vaping could avert 6.6 million premature deaths.
With all the obvious benefits e-cigarettes confer, government officials should be very cautious about how they regulate or communicate about these potentially lifesaving products. Anything that makes e-cigarettes less attractive to smokers will result in fewer smokers switching and more smokers dying. If the law, as it currently exists, goes into effect that is exactly what will happen. Congress should step in now and correct this grave error, before it is too late.
Read previous posts in the “Last Chance for the 115th” series:
- Reforming the Renewable Fuel Standard by Ben Lieberman (6/21/18)
- Keeping the Internet Sales Tax at Bay by Jessica Melugin (6/20/18)
- Options for Regulatory Reform by Ryan Young (6/19/18)
- Unshackle Middle-Class Investors and Entrepreneurs by John Berlau (6/18/18)
- Labor and Employment by Trey Kovacs (6/15/18)
- Stop the President from Unilaterally Raising Tariffs by Ryan Young (6/14/18)
- Bring Accountability to the Financial Regulators by Daniel Press (6/13/18)
- Senate Should Pass AV START Act by Marc Scribner (6/12/18)
(This post has been updated.)